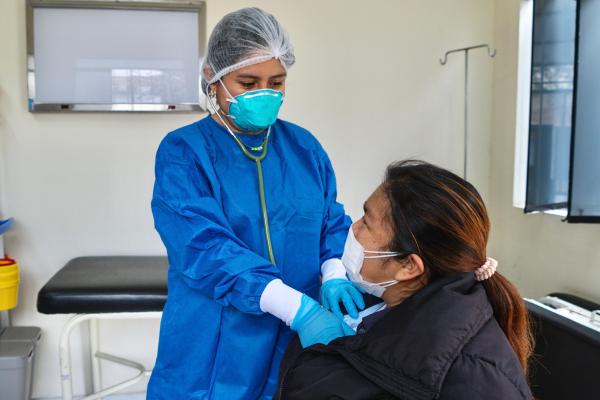
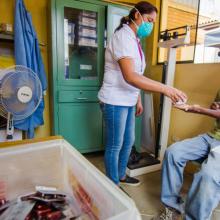
Being a patient with tuberculosis (TB) is an exhausting experience. About 52,000 people who contract this disease in Peru each year know this well and, in addition to suffering its social stigmas and economic cost, must receive harsh treatment.
Curing a typical form of TB requires four to six months of therapy, but if someone gets multidrug-resistant (MDR-TB) or extensively drug-resistant (XDR-TB), then they can spend 18 to 24 months taking up to 14,000 pills and receiving painful daily injections.
This difficult scenario, however, is nearing its end.
The answer is called endTB. A consortium led by Partners In Health, Interactive Research and Development and Médecins Sans Frontières, and funded by Unitaid, which over the past decade has tested five new, all-oral, shortened treatment regimens for MDR-TB lasting only nine months.
This is not, of course, a miracle. This result is the fruit of a long collaboration with several scientific studies at different stages, in which Partners In Health (SES) has played a crucial role, as have several institutions around the globe. Therefore, the following is a story of hope in modern science, but also in human bonds.

El consorcio endTB consiguió acortar a nueve meses el tratamiento para pacientes con TB-MDR, así como eliminar los inyectables que volvían más doloroso el proceso de recuperación.
Two Drugs, One Path
It was 2012 when the U.S. Food and Drug Administration (FDA) approved the use of bedaquiline to treat TB. It had been nearly 50 years since the last time the U.S. agency had given the green light to a new drug to treat the disease.
Just two years later, the European Union did the same with a new drug: delamanid. “We had two drugs to treat resistant tuberculosis, but for various issues no country implemented it,” says Dr. Leonid Lecca, general director of Socios En Salud (SES) and instructor at Harvard University.
Faced with that scenario, endTB set out to act under two research arms: one observational and the other experimental. The first, according to Lecca, sought to advocate and accompany at the institutional level so that each country would integrate these two new drugs into their standard treatment schemes for multidrug-resistant forms of tuberculosis.
By those years, SES met with various Peruvian health authorities to “achieve authorization for the use of the new drugs,” as Antonieta Alarcón, now executive director of the Directorate for Tuberculosis Prevention and Control (DPCTB) of the Ministry of Health (MINSA), recalls.

El monitoreo y seguimiento fueron pasos claves durante el desarrollo de la iniciativa internacional endTB, que tuvo casi un 40% de participantes de nacionalidad peruana.
Thanks to this initiative, Peru became the first Latin American country to introduce bedaquiline in the treatment of TB. A milestone that was soon followed by the entry of the first batches of delamanid in our country, as well as the reintroduction of a drug that had ceased to be used at the beginning of the new century: clofazimine.
“We started the intervention with part of these drugs and this operational arm of endTB in eight hospitalized patients with XDR-TB, to whom another treatment was administered intravenously (…) We greatly reduced the percentage of deaths, to the point that now the rate is between 17% and 18%, and we reached almost 80% cure rate,” says Alarcón.
With 267 people enrolled in the observational study, endTB had achieved one of its objectives, but it still had a long way to go: the experimental one. “We not only wanted to show that the drugs improved the cure rate, but also that shorter schedules improved healing and people’s experience,” Lecca stresses.
endTB set out to advocate and accompany at the institutional level for each country to integrate new drugs into their standard treatment schemes for multidrug-resistant TB.
A global effort: the endTB clinical trial
In 2017, endTB launched its five-arm clinical trial of schemes to reduce the duration of TB treatment. The initiative enrolled 754 patients in seven countries on four continents: Peru, Georgia, Lesotho, Pakistan, South Africa, India and Kazakhstan. Nearly 40% of the participants were Peruvian nationals.
“To choose the countries, it was necessary for them to have a significant burden of MDR-TB,” says Carole Mitnick, PIH research director for the endTB project, co-principal investigator of the study and professor of global health and social medicine at the Blavatnik Institute at Harvard Medical School.
Mitnick notes that they also sought “a diverse group of participants,” since people with MDR-TB have “significant comorbidities, such as HIV, diabetes, alcohol and drug use.” “In clinical trials, they usually exclude somewhat complicated participants so as not to threaten good outcomes, but for us these are part of the group of patients we want to help,” he stresses.

La doctora Carole Mitnick, directora de investigación de Partners In Health para el proyecto endTB, co-investigadora principal del estudio, y profesora de Medicina Global y Medicina Social en la Facultad de Medicina de Harvard, durante la presentación de resultados del ensayo clínico endTB.
In Peru, patients from the Hipolito Unanue, Dos de Mayo and Sergio E. Bernales national hospitals participated in the clinical trial, which was supported by researchers and physicians from Harvard Medical School and other research centers around the world.
“The five arms of the clinical trial were nine months long and all used oral medications. In addition to other drugs, two of these arms included bedaquiline, two others delamanid, and one arm bedaquiline and delamanid. Clinical trial enrollment ran from February 2017 to October 2021,” Lecca comments.
Thus, in October 2023, endTB presented its results at the latest edition of the Union World Conference on Lung Health, held in Paris (France): three of the five arms evaluated in the clinical trial achieved effectiveness and high cure rates (~85%), as well as shortening treatment for MDR-TB and freeing it from injections.
New hope was dawning for the half a million people each year who are infected with this form of TB around the world.
MINSA implements shortened endTB schemes
Far from France, another milestone occurred between October and December 2023 in Peru. The endTB had shared its results with MINSA, and the latter was quick to implement in 46 MDR-TB patients the new oral and shortened treatment scheme proposed by arm two of the clinical trial. The therapy comprises the intake of bedaquiline, clofazimine, linezolid, levofloxacin and pyrazinamide for nine months.
According to Alarcón, the fact that almost 40% of the participants in the clinical trial were Peruvians was “strong evidence for us, as a country, to be able to introduce this type of drugs.”
According to Alarcón, the fact that almost 40% of the participants in the clinical trial were Peruvians was “strong evidence for us, as a country, to be able to introduce this type of drugs. “The implementation was in Lima and Callao, above all, because it is closer to us to work in one way or another these processes,” he indicates.
“It has helped us a lot to shorten the treatment schemes. The consultant pulmonologists who have participated in the endTB process have found it very easy to adopt it, and I think it is something we urgently needed. It was a demand from patients to stop using injectables,” says the MINSA representative.
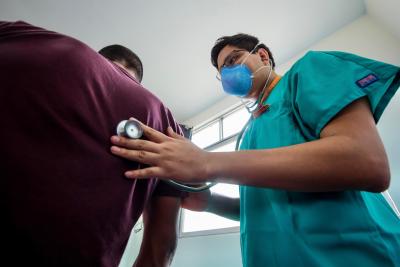
Socios En Salud colaboró con el Ministerio de Salud para llevar a cabo la implementación del nuevo régimen de tratamiento para pacientes con TB-MDR, propuesto por el brazo dos del ensayo clínico del endTB.
Unlike other short-course therapies, Alarcón stresses that the regimens proposed by endTB can be applied in patients from 0 years of age, with comorbidities and even in pregnant women. “This 2024 we already have 66 more patients recruited. There are a total of 112 who have started treatments with shortened oral schemes,” he reports.
Branch one of the endTB clinical trial is also being implemented in Peru. The DPCTB executive director expects that with these regimens, treatment dropout rates will decrease and “adherence rates” will increase. By the end of March they will have “a cohort of the first ones who entered in October to analyze bacteriological conversion.”
Meanwhile, MINSA has a pending task: to update the technical health standard for the comprehensive care of the person affected by tuberculosis, family and community. “We are including (these regimens) within the new version, mainly arm two and one, which will be written as proposals,” Alarcón points out.
Also, who heads the DPCTB states that the government has already ordered the purchase of more drugs to expand the shortened endTB treatment to other regions of Peru. “The country is grateful for these boosts to research that allow us to make decisions and changes in public policy,” he says.
The treatment schemes proposed by endTB can be applied in patients as young as 0 years old, with comorbidities and even in pregnant women.
The endTB in the WHO
Starting in 2018, says the CEO of Partners In Health, the World Health Organization (WHO) placed bedaquiline and delamanid, in addition to other drugs, among the medications that should be used in therapies for the different forms of tuberculosis. “Since that year, it recommends no longer using injectables,” says Lecca.
Currently, according to Carole Mitnick, Partners In Health (PIH) is collaborating with the WHO so that next June its findings on endTB will be considered for inclusion in the global guidelines for treating multidrug-resistant TB.
“This step is important because many countries rely on WHO recommendations to prepare their own guidelines. In addition, donors such as the Global Fund require that grant activities be endorsed by WHO,” she notes.
Although all of these initiatives are giant steps toward fighting the spread of Koch’s bacillus, the Harvard Medical School professor emphasizes that there are still barriers. “One of them is the cost of treatment,” she warns. That’s why PIH is already working to convince the pharmaceutical industry to release patents on certain drugs.
But in the meantime, millions of people may see the light at the end of the tunnel.